Solectrix Q&V: Regulatory Excellence from Practical Experience for Practice Use
As a long-standing development and production service provider, we at Solectrix know: the path from the initial idea to an approved and successful medical device is long and accompanied by ups and downs.
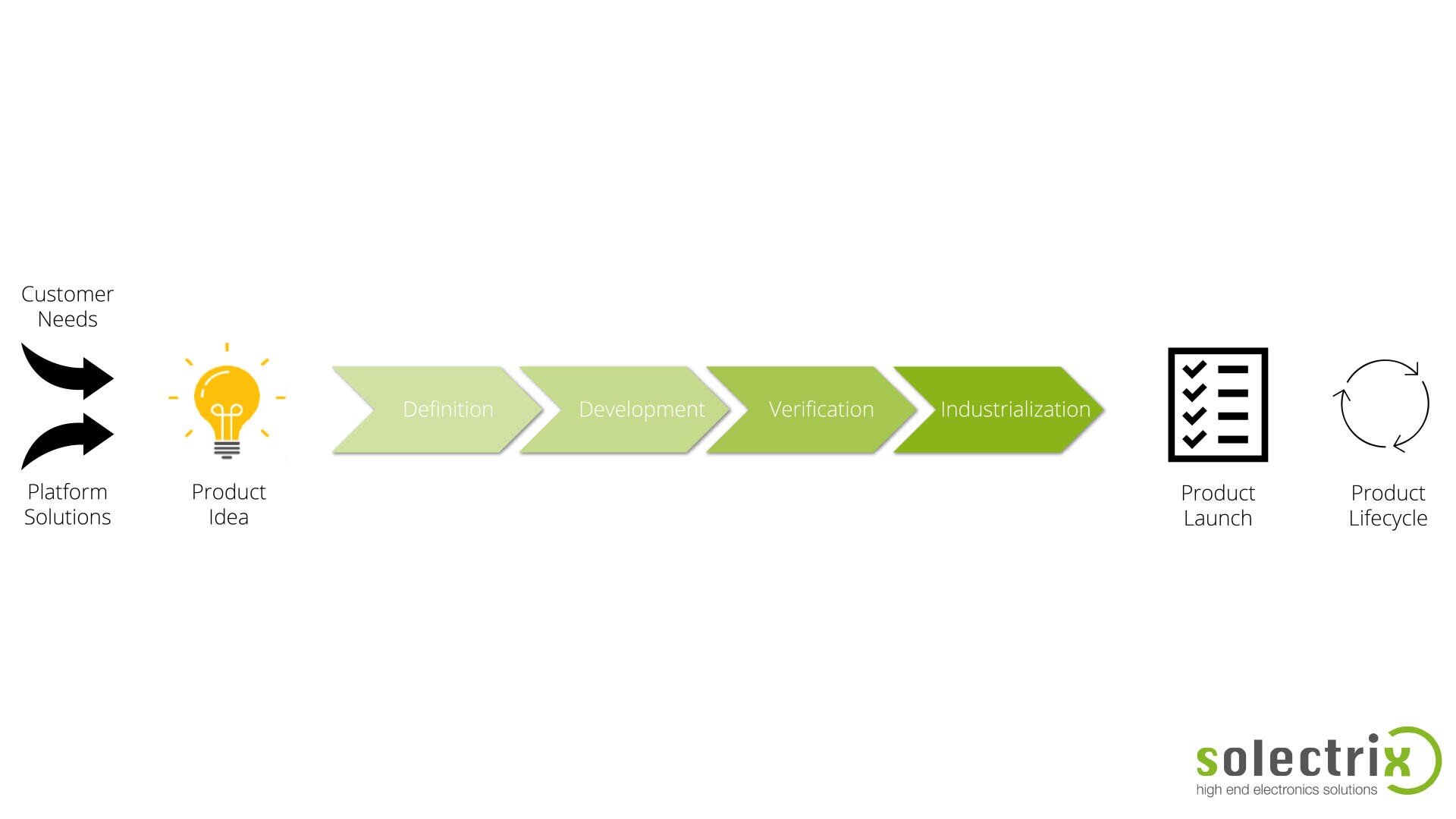
Your Partner for the Entire Medical Technology Product Life Cycle
As a long-standing development and production service provider, we at Solectrix know: the path from the initial idea to an approved and successful medical device is long and accompanied by ups and downs. In this, regulatory requirements are not obstacles, but guidelines that ensure safety, quality, and sustainability.
That is precisely why our Qualification & Verification (Q&V) department at our Mannheim location is more than just a consultant – it is your partner, combining the theory of norms with the daily practice of product creation. Our expertise does not come from studying the literature, but has been developed over countless development projects. We offer you practical, tried-and-tested support to bring your innovations to market safely, efficiently, and sustainably.
Our Holistic Approach: Quality from Idea to Market Monitoring
We accompany you through every phase and ensure that regulatory, normative, and qualitative aspects are an integral part of your project from the start.
Phase 1: Strategy & Concept – Quality from the Outset (“Quality by Design”)
The greatest lever for success lies in the early phase. We help you set the right course from the first minute to avoid costly and time-consuming redesigns and delays. This includes developing a viable certification strategy and establishing a central risk management process in accordance with ISO 14971, which runs like a thread through the entire project.
Phase 2: Development & Documentation – Conformity in Detail
We put the strategy into practice. Our experts actively support you in creating robust technical documentation in accordance with MDR/IVDR. A particular focus is placed on usability (Usability & Human Factors Engineering according to IEC 62366), because a safe product must above all be safe to use. Since we develop ourselves, we know how to create documentation efficiently, comprehensibly, and in line with development.
Phase 3: Verification, Validation & Testing – Objective Proof
We provide proof that your product meets the requirements – safely, reliably, and reproducibly. We plan and manage all V&V activities, perform Computer Software Validation (CSV), and coordinate EMC testing (IEC 60601-1-2) with accredited labs. Our goal is not only to pass the tests, but to provide robust proof of your product's quality.
Phase 4: Production & Supply Chain – Assured Process Quality
The quality of the product depends directly on the quality of its manufacturing. We ensure this through systematic equipment qualification (IQ, OQ, PQ). In addition, we contribute our expertise as a production service provider and support you in the strategically important selection and qualification of your suppliers. This way, we create a robust and compliant supply chain.
Knowledge Transfer: Training Courses & Workshops
We share our practical experience with you. In individual workshops and practical training courses on topics like technical documentation, risk management, MDR/IVDR, and usability, we enable your team to build up regulatory expertise and anchor it firmly in your company.
Solectrix Q&V is your integrated solution partner. We combine the strategic foresight of a consultant with the pragmatic implementation expertise of a developer and manufacturer.
Talk to us! Let's work together to make your project a success.